【深圳大學:增效型水凝膠電解質,實現能量自主和可穿戴傳感】
傳統的熱電轉換技術以熱電發生器為特征,利用基于固態半導體的電子塞貝克效應來實現熱流到電流的直接轉換。然而,這些熱電發生器通常具有剛性、毒性或熱功率低(約為 uV K-1)的缺點,這阻礙了它們在可穿戴電子設備中的進一步發展。相比之下,熱電偶電池(TEC,又稱熱電化學電池或熱電偶電池)可在室溫下產生較大的熱功率,因此特別適用于收集人體和可穿戴電子設備的低品位廢熱。在溫度梯度下,一個電極發生氧化反應,另一個電極發生還原反應。這些連續的氧化還原反應導致電子在外電路中不斷流動,并在冷熱電極之間產生電位差,從而產生很大的熱電化學塞貝克系數(Se),其數量級為mV K-1。
為了推動柔性和可穿戴應用的發展,近期有關 TEC 的研究越來越多地將重點轉向水凝膠電解質。對于柔性設備來說,水凝膠電解質可以避免傳統液基 TEC 的電解質泄漏和復雜封裝問題。然而,柔性和可穿戴 TEC 的開發面臨著一些挑戰,其中最主要的是如何提高其熱電化學性能。提高熱功率以實現高效率對于最大限度地利用低品位熱源進行能量轉換至關重要。在提高熱功率的同時,還面臨著提高機械強度的挑戰,特別是對于基于水凝膠的 TEC 而言,因為它們必須具備強大的機械性能,以承受撕裂、彎曲和扭曲。這一點在可穿戴應用中尤為重要,因為在這些應用中,靈活性和耐用性是不可或缺的,而目前的研究卻在很大程度上忽視了這一點。除了熱功率低和機械強度不足外,目前的準固態 TEC 還缺乏在受到嚴重外部損壞后恢復的能力,這大大限制了它們在柔性和可穿應用中的實用性。
研究成果
柔性準固態熱電偶(TEC)的快速發展為可穿戴電子設備提供了一條嶄新的發展道路。然而 機械強度和功率輸出的不足仍然阻礙著它們的進一步應用 。深圳大學陳光明教授團隊研究展示了一種“一石二鳥”策略,可協同增強基于[Fe(CN)6]3-/4-的熱電偶的機械和熱電化學特性。通過甜菜堿齊聚物引入多種非共價相互作用,傳統脆性明膠水凝膠電解質的機械強度從50 kPa 大幅提高到440 kPa,伸展性接近 250%。同時,甜菜堿齊聚物強烈影響了[Fe(CN)6]3-的溶解結構從而擴大了熵差,并將熱電化學塞貝克系數從1.47 mVK-1提高到2.2 mV K-1。由此產生的準固態 TEC 顯示出0.48 mW m-2 K-2的超高歸一化輸出功率密度,與未進行齊聚離子調節的同類產品相比,整體性能有了顯著提高。此外,固有的熱可逆特性使TEC能夠通過溶膠轉化反復進行自我恢復,從而確保可靠的能量輸出,甚至在發生極端機械損壞的情況下也能對 TEC 進行回收利用。作者還進一步設計了一種由18個獨立TEC組成的能量自主智能手套,它可以同時監測任何被觸摸物體不同位置的溫度,在可穿戴應用中展現出巨大的潛力。
圖文導讀
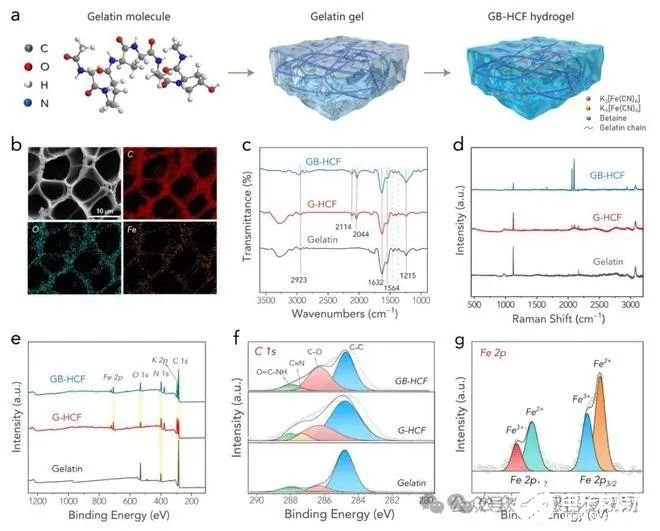
Figure 1. (a) An illustration of the forming process of the GB-HCF hydrogel. (b) An SEM image of the GB-HCF and the corresponding elemental mapping images. (c) FTIR spectra of various samples. (d) Raman spectra of various samples. (e) XPS full spectra of various samples. (f) XPS C1s spectra of various samples. (g) XPS Fe2p spectrum of the GB-HCF.
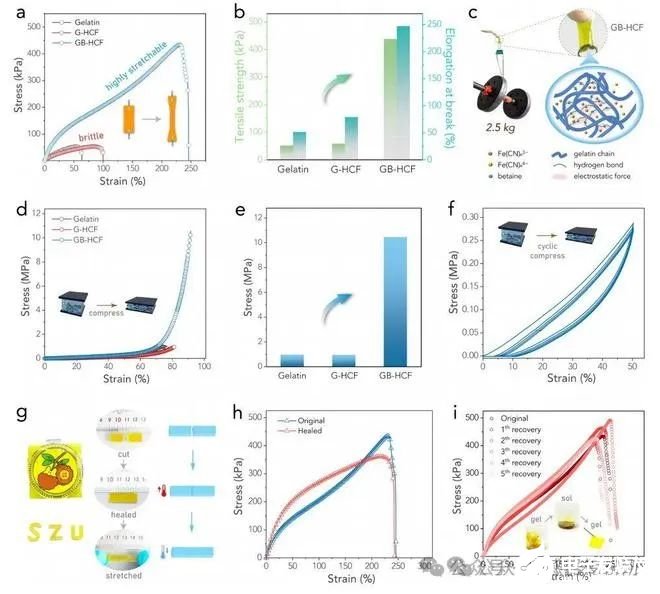
Figure 2. (a) Stress-strain curves of various samples. (b) The corresponding tensile strength and elongation at break of various samples obtained from (a). (c) Demonstration of the GB[1]HCF hydrogel lifting a weight of 2.5 kg. The schematic diagram highlights the role of betaine in the hydrogel network, where the betaine molecules can bind the gelatin chains tightly through multiple non-covalent interactions including hydrogen bonds and electrostatic interactions. (d) Compression curves of various samples. (e) The corresponding compressive strength of various samples obtained from (d). (f) Stress-strain curves of cyclic compression of the GB-HCF. (g) Demonstration of the tailorable GB-HCF hydrogels and their healing capability. (h) Stress[1]strain curves of the GB-HCF before and after healing. (i) Stress-strain curves of the GB-HCF subjected to five recovery cycles. Insets show its self-recovery process based on the sol-gel transformation.
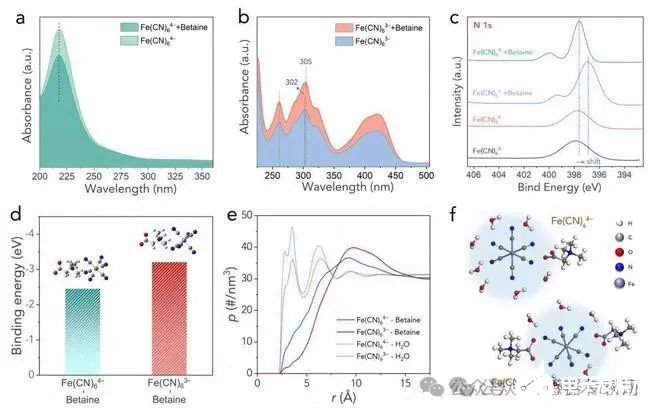
Figure 3. (a) UV-vis spectra of the [Fe(CN)6]4? and [Fe(CN)6]4? + betaine sample. (b) UV-vis spectra of the [Fe(CN)6]3? and [Fe(CN)6]3? + betaine sample. (c) XPS N 1s spectra of various samples. (d) The calculated binding energies for [Fe(CN)6]4-betaine and [Fe(CN)6]3?-betaine. Insets show the optimized binding structures. (e) The radial density profiles between [Fe(CN)6]3?/[Fe(CN)6]4? and water/betaine molecules. (f) The illustration of the solvation structures of [Fe(CN)6]3? and [Fe(CN)6]4? with the presence of betaine.
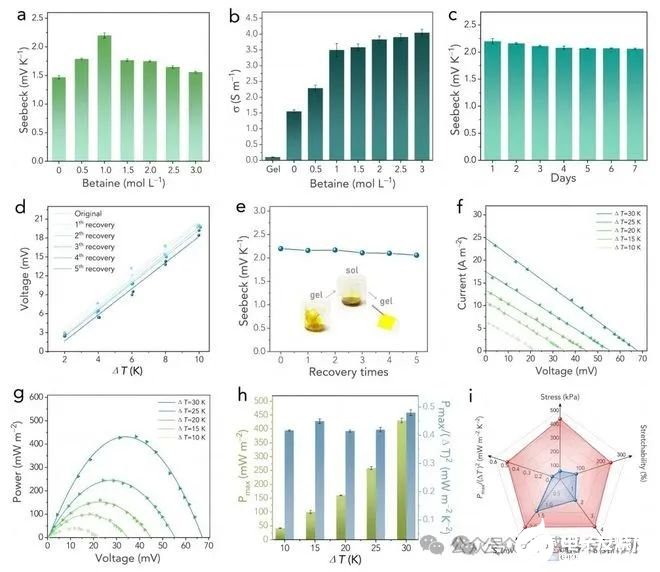
Figure 4. (a) The effect of betaine concentration on the Se. (b) The effect of betaine concentration on the ionic conductivity. (c) The Se change during the rest in air for 7 days. (d)The fitted plots of voltage-temperature difference for different recovery cycles. (e) The measured Se values after different recovery cycles. Insets show the recovery cycle. (f) Current density-voltage plots under different ?Ts. (g) Power density-voltage plots under different ?Ts. (h) The calculated maximum specific output power density. (i) Comprehensive comparisons between the GB-HCF based TECs and conventional gelatin-based TECs in terms of Pmax/?T2, Se, σ, tensile strength, and stretchability.
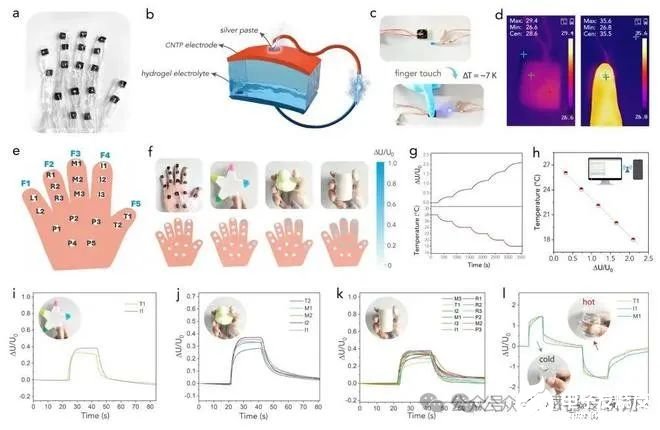
Figure 5. (a) An optical image of the smart glove consisting of 18 TEC blocks. (b) Schematic of the configuration of a single TEC block. (c) Photos showing a TEC block powering a light[1]emitting diode bulb by utilizing the body heat with the assistance of a voltage amplifier. (d) Corresponding infrared thermal images. (e) A schematic of the hand-shaped smart glove. (f)Demonstration of wearing the smart glove device to hold different objects. The schematics in the lower part show the voltage responses in different zones of the hand. (g) The variation of ?U/U0 values with the variation of the target temperature. (h) The relationship between the ?U/U0 values and the target temperature. (i-l) The voltage responses of the device when the hand was touching a pentagram toy, a duck toy, a cylinder, and a cold/hot water cup. Insets are the photos showing how the hand was holding the objects.
聲明:轉載此文是出于傳遞更多信息之目的,若有來源標注措誤或侵犯了您的合法權益,請與我們聯系,我們將及時更正、刪除,謝謝。
-
電解質
+關注
關注
6文章
833瀏覽量
21432 -
傳感技術
+關注
關注
5文章
561瀏覽量
47755 -
可穿戴傳感器
+關注
關注
2文章
103瀏覽量
13142
發布評論請先 登錄
高成本難題破解:新型非晶態鹵化物固態電解質引領行業變革

國內企業突破固態鋰電池電解質瓶頸
全固態鋰電革命:垂直取向超離子通道復合電解質的創新突破

全北國立大學:研究可穿戴汗液傳感貼片,用于無創連續健康監測

MLPC的抗振性能如何與液態電解質電容比擬

深圳大學:研發新型傳感器,實現阿摩爾級生物分子精準檢測
巴西研究團隊推進鈉離子電池電解質計算研究

突破性固態聚合物電解質:像拼圖一樣組裝分子,打造安全高壓鋰電池

安徽大學:基于聚乙烯醇/絲素蛋白/石墨烯水凝膠的柔性傳感器,用于先進的可穿戴電子產品等
鋰離子電池電解質填充工藝:技術原理與創新實踐

國儀電鏡助力PANC/T-Fe水凝膠在不同環境溫度下的微觀結構分析
水凝膠拉伸試驗機:材料性能的洞察者




 深圳大學:增效型水凝膠電解質,實現能量自主和可穿戴傳感
深圳大學:增效型水凝膠電解質,實現能量自主和可穿戴傳感




評論